Arthrosamid injections
Effective, long-lasting pain relief from osteoarthritis in the knee, without the need for surgical intervention.

What is Arthrosamid® ?
Change the color to match your brand or vision, add your logo, choose the perfect layout, modify menu settings, add animations, add shape dividers, increase engagement with call to action and more.
Arthrosamid is the first and and only approved injectable treatment that permanently combines with the knee’s synovial tissue.
The hydrogel injection is administered by ultrasound guidance and eases the symptoms of knee osteoarthritis. The hydrogel itself does not degrade and therefore aims to provide long-lasting pain relief and improve your quality of life.
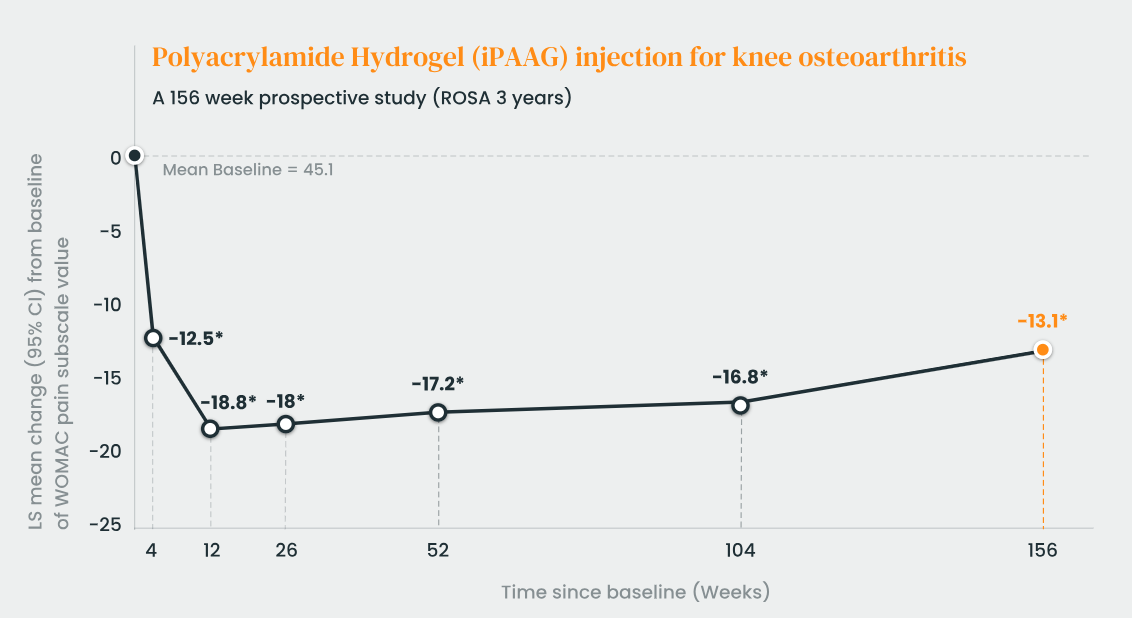
Arthrosamid® is proven to maintain a significant, long-lasting reduction in knee OA pain even three years post-treatment.
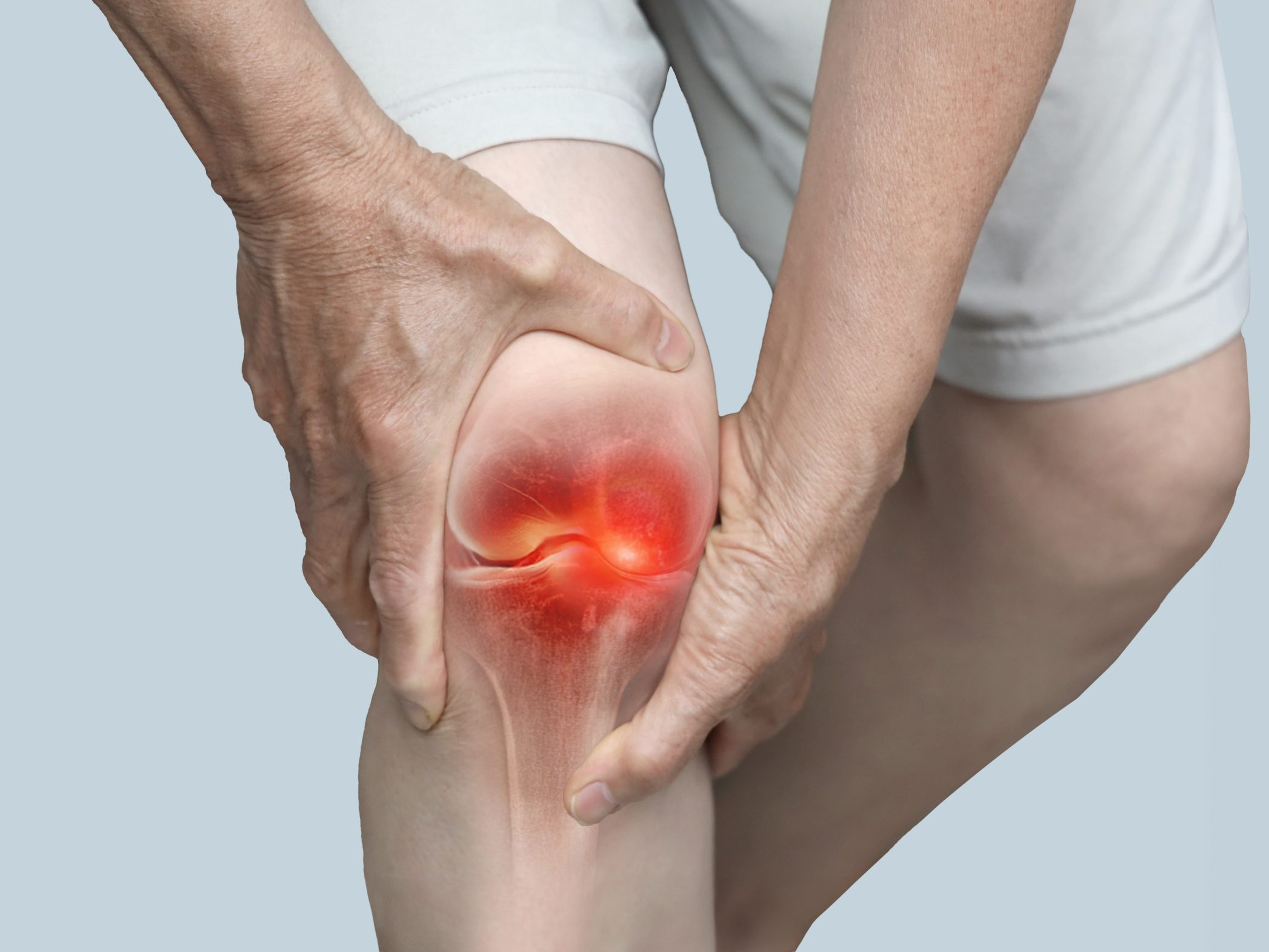
What is Knee Osteoarthritis?
Knee osteoarthritis (OA) is a long-term condition where the shock-absorbing cartilage is worn away, causing bones to rub together and the joint to become stiff, swollen and painful. The knee joint worsens over time, resulting in synovial pain and disability.
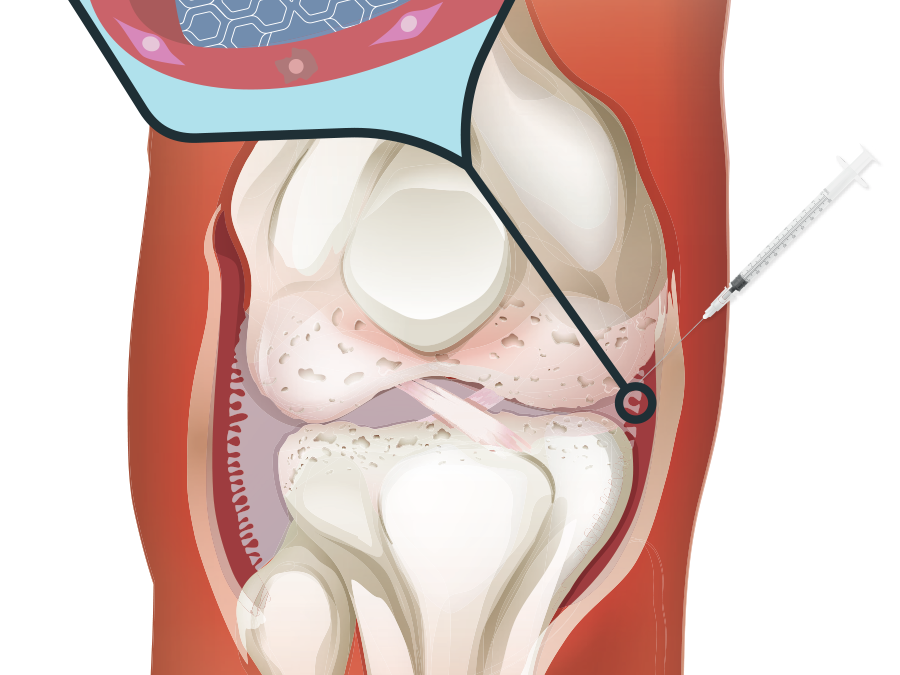
How is Arthrosamid ® Administered?
Before treatment with Arthrosamid®, you should be given antibiotics ahead of this to protect you from any potential risk of infection.
Your knee is cleaned prior to treatment. Arthrosamid® is then injected into your knee under ultrasound guidance to ensure pinpoint accuracy. The needle is then removed and a plaster is placed over the injection site.
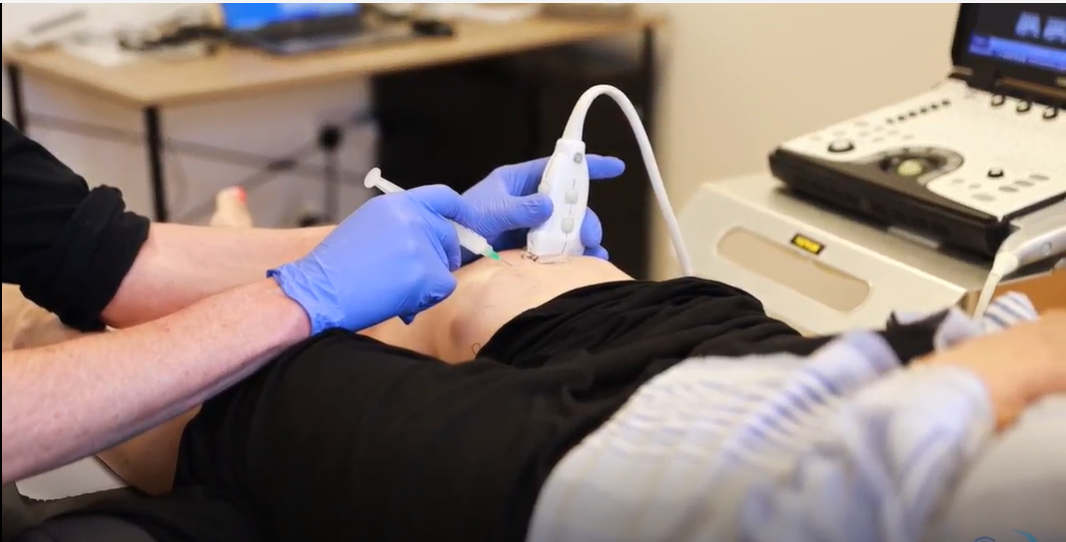
What is the evidence for Arthrosamid's beneficial effects?
What is the evidence for Arthrosamid's beneficial effects?
Although large-scale, long-term randomised studies have not yet been published, several smaller studies and observational reports provide insight into Arthrosamid’s performance. Improvements of around 30–40% have been reported at follow-up intervals ranging from 6 to 12 months. Previously reported data from the prospective open-label study “IDA” had demonstrated Arthrosamid’s effect in reducing pain at 6 months, which was maintained at 12 months (1).
ROSA study: The aim of the study was to investigate the long-term efficacy and safety of a single injection of 6ml intra-articular polyacrylamide hydrogel (iPAAG, Arthrosamid®) on knee symptoms in participants with moderate to severe knee osteoarthritis (OA) for up to 5 years after treatment and to evaluate the time to start other treatments for knee OA. In this IDA study, 49 patients with knee osteoarthritis received a single injection of 6 mL of a 2.5% polyacrylamide hydrogel. Over a follow-up period of three years, there were statistically significant and clinically relevant improvements in key measures—including the WOMAC subscale scores for physical pain, stiffness, and function.
- Bliddal H, Overgaard A, Hartkopp A, Beier J, Conaghan PG, et al. (2021) Polyacrylamide Hydrogel Injection for Knee Osteoarthritis: A 6 Months Prospective Study. J Orthop Res Ther 6: 1188.
- Bliddal, H., et al. (2024) 3 year follow-up from a randomized controlled trial of intra-articular polyacrylamide hydrogel injection in subjects with knee osteoarthritis. Osteoarthritis and Cartilage. Vol 32 (6): 770-771.
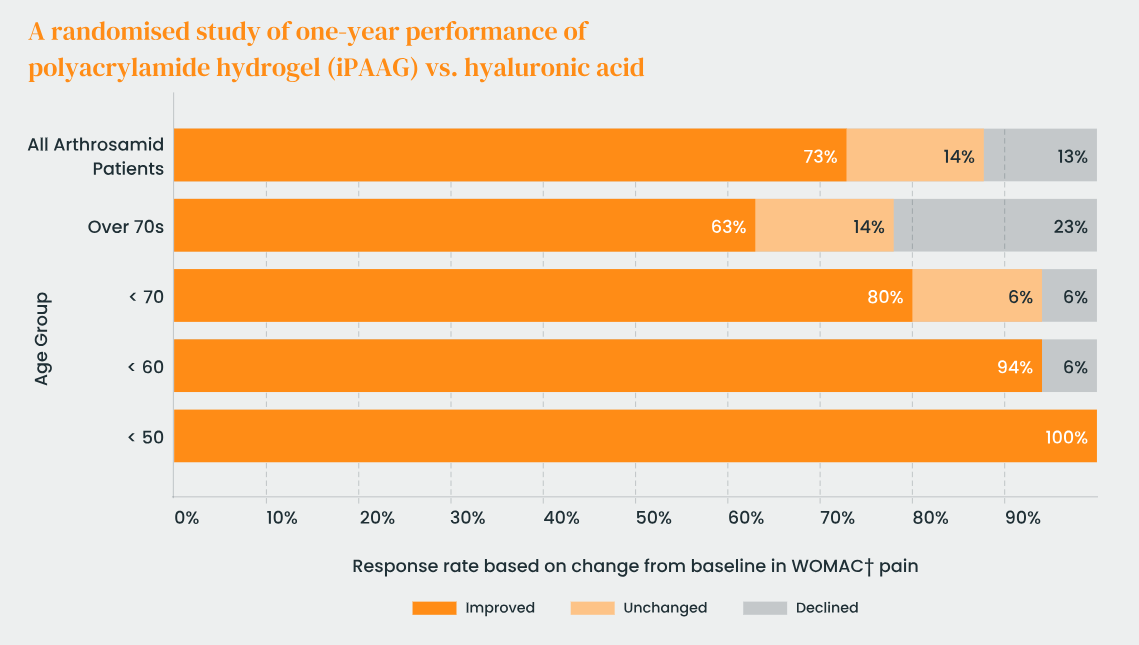
What is the the response rate with Arthrosamid ® ?
Approximately 60–70% of osteoarthritic knee patients treated with Arthrosamide achieve a clinically meaningful improvement compared to baseline. In contrast, placebo groups in similar trials have sometimes shown improvements in the range of 35–40%, highlighting a treatment effect. The response was typically defined as a reduction in pain scores by at least 30% and a similar or greater improvement in functional scores (e.g., WOMAC). These thresholds are commonly accepted as the minimal clinically important difference in OA.
Larger trials will refine these response rate estimates and help better predict which patients are most likely to benefit.
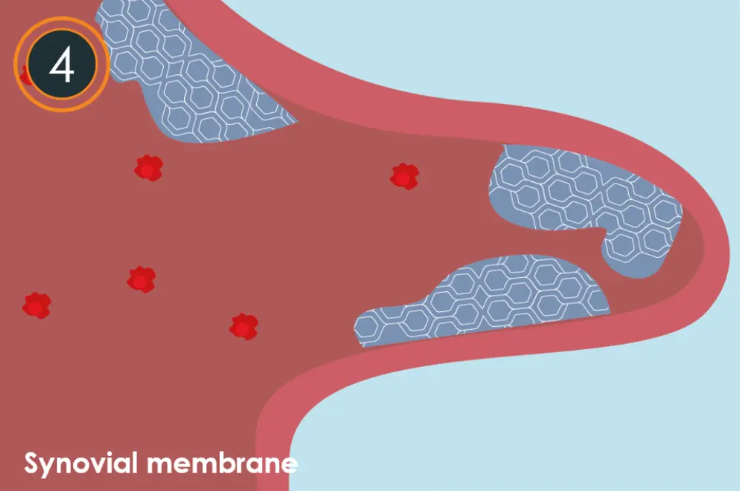
How does Arthrosamid ® work?
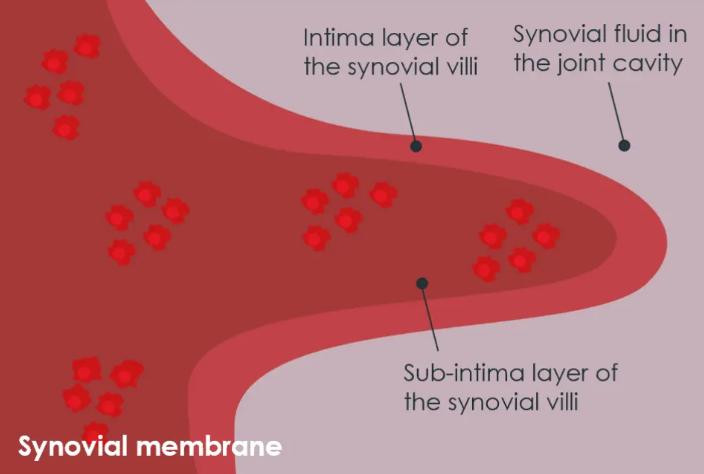
Image left top: Untreated OA knee: In a knee affected by OA, the synovial fluid loses its viscoelastic properties, which are crucial for joint lubrication and shock absorption. This loss leads to a fibrotic synovial membrane which in turn causes an accumulation of inflammatory cells. These inflammatory cells are known to be a precursor to pain and swelling.
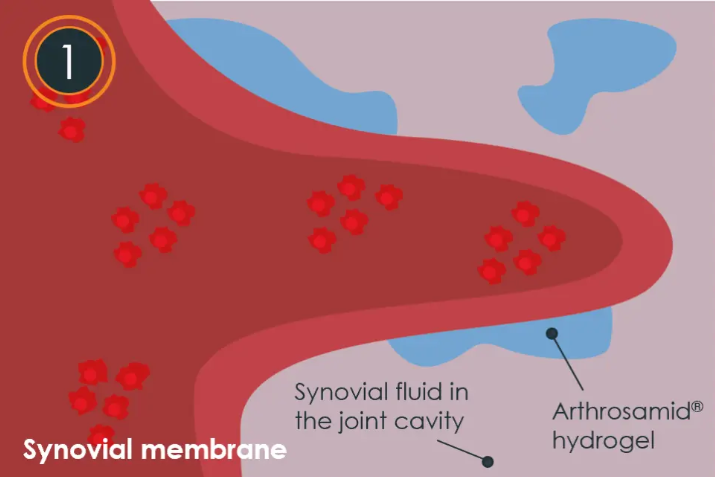
- Delivered with a single injection Arthrosamid® is injected into the synovial cavity, where it disperses within the synovial fluid and adheres to the synovial membrane through the tissue’s normal ‘filtering’ mechanisms.
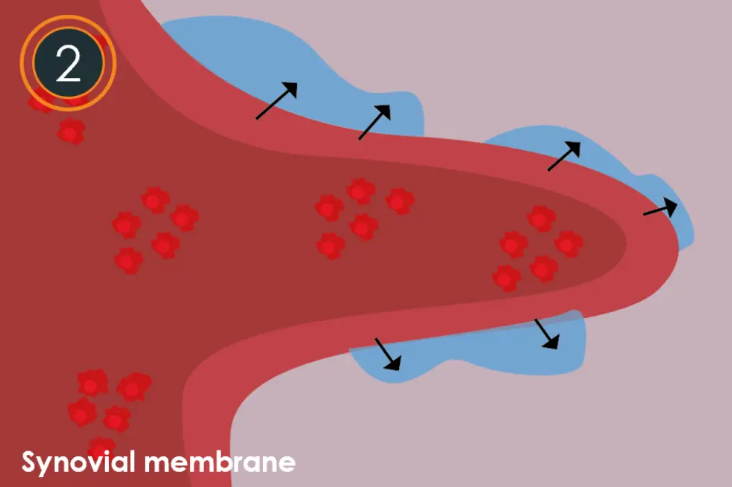
- Arthrosamid ® is integrated with the synovial membrane The synovial membrane begins to incorporate the non-biodegradable polyacrylamide hydrogel (2.5% iPAAG) via its natural uptake mechanism.
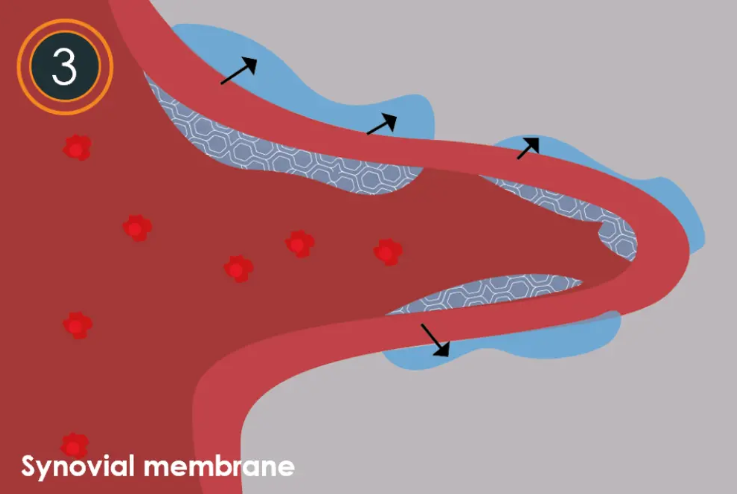
- Step 3: Improved knee joint function As 2.5% iPAAG becomes integrated, it begins to soften and/or increase the elasticity of the fibrotic synovial membrane leading to clinically proven decreased joint stiffness and pain, and improved knee function.
- Step 4: Complete integration Within approximately 6-12 weeks, the injectable 2.5% iPAAG implant fully integrates into the synovial membrane acting as a synovial scaffold within the subintima layer.
Ready to take the next step?
If your pain has not improved with regular management strategies such as physiotherapy and regular pain medication, and you have only had limited benefit from regular types of injection therapy such as steroid injections, then it may be helpful to talk to us about Arthrosamid®.